Checking the functioning of the HydroFlex
Ideally, before your actual measurements you check the hydrogen electrode against a so-called master electrode, e.g. a calomel electrode. The master electrode should always stand in suitable potassium chloride solution and should not be used for other purposes. Mind the fill level of the calomel electrode which always has to be above the liquid level in the measuring vessel. Open the filling plug of the calomel electrode to pretend the measuring liquid from entering into the calomel electrode.
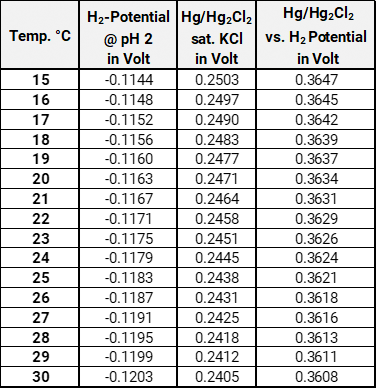
To control the hydrogen electrode, we suggest to put the hydrogen and the calomel electrode in buffer pH 2. Connect the calomel electrode with the positive pole and the hydrogen electrode HydroFlex with the negative pole (Com-port) of your voltmeter. After an adjustment phase you can read off the potential. Note that diffusion voltages set at the calomel electrode. The measured values can be higher (approx. 5 mV) than the ideal values in the table below.
download Potential table in Buffer pH 2Hydrogen potential, potential of the calomel electrode and the resulting measuring potential for temperatures between 15°C and 30°C in buffer pH 2. The figures for the H2-potential are calculated with the Nernst equation. The figures of the calomel electrode are taken from D. Dobos, Electrochemical Data, 1975, Elsevier.



 Deutsch
Deutsch